|
Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. 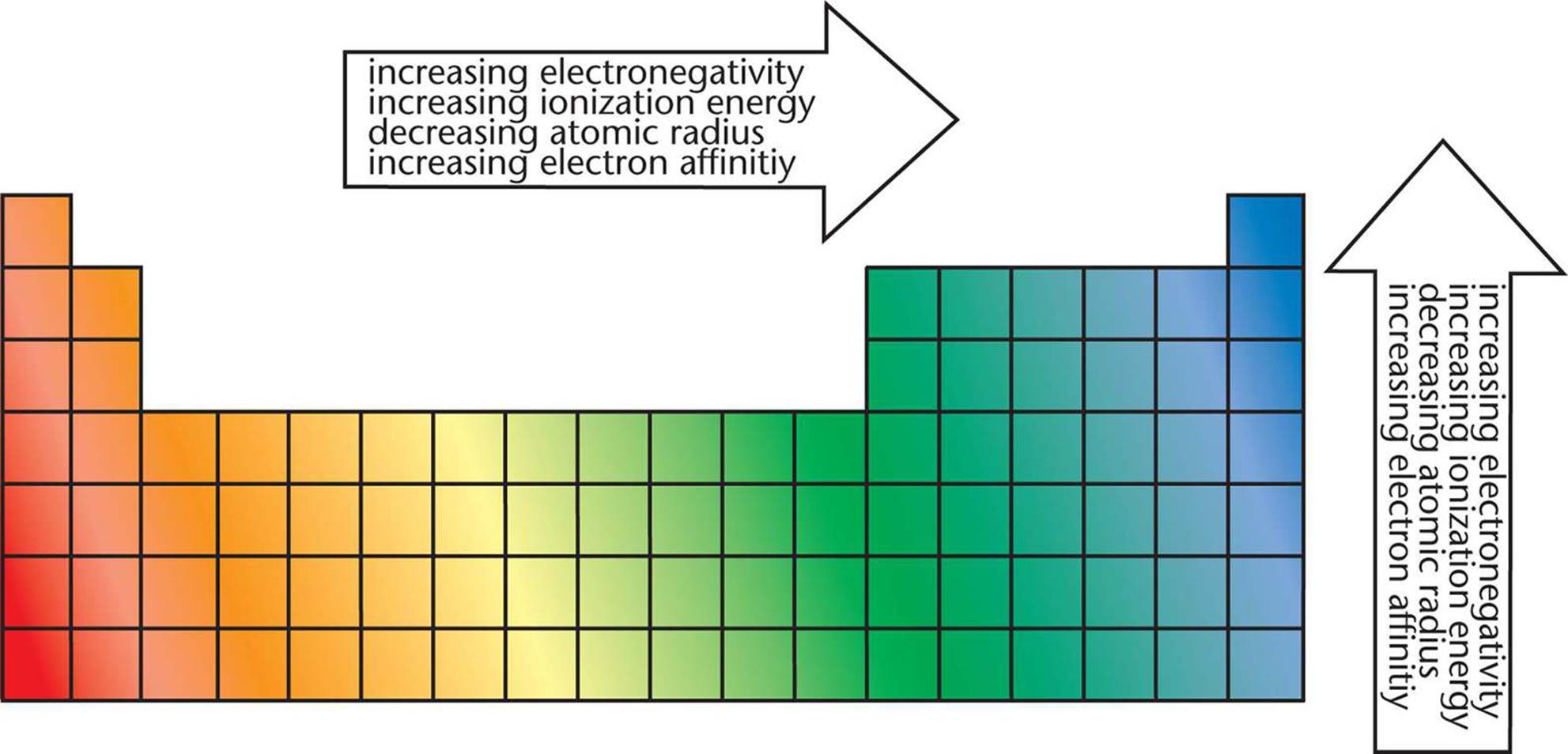
As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. First, electrons are added one at a time moving from left to right across a period. In addition to this activity, there are two other important trends. Stable octets are seen in the inert gases or noble gases, of Group VIII of the periodic table. These trends can be predicted using the periodic table and can be explained and understood by analyzing the electronic configurations of the elements.Įlements tend to gain or lose valence electrons to achieve stable octet configuration. The properties of the elements in a periodic table exhibit trends. T True or False? Atomic radii decreases from left to right.Periodic trends in properties of elements T True or False? Atomic radii increases from top to bottom. True or False? Ionization energy decreases from left to right. T True or False? Ionization energy decreases from top to bottom. 
True or False? Electron affinity increases from top to bottom. T True or False? Electron affinity increases from left to right. Group: As there is strong correlation between atomic radii and atomic radii increases, electronegativity decreases as the nucleus of a large atom is much further away to the valence electrons compared to a small atom. The nucleus of a small atom can get much closer to the valence electrons of another atom than the nucleus of a larger atom can. Period: There is correlation between atomic radii, therefore the smaller the atom, the larger its electronegativity. Trends in Electronegativity Increases from left to right, decreases from top to bottom. Nuclei of both atoms attract shared electrons. Electronegativity A number that describes an atom's ability to attract a bonding atoms' electrons. True or False? If energy is absorbed when an electron is added, electron affinity is low and is expressed as a positive integer. True or False? If energy is released when an atom of an element gains an electron, the electron affinity is expressed as a positive integer. F, it is expressed as a negative integer if energy is released when an atom of an element gains an electron. Electron Affinity Equation A(g) + e- → A+(g) + energy T True or False? The trends for the elctron affinity are the same as for the trends in ionization energy. Electron Affinity The energy absorbed or released when an electron is added to a neutral atom in the gaseous state. True or False? The higher the first IE, the more reactive (excluding the noble gases) a nonmetal is. The more difficult it is to remove the 1st electron, the easier it will gain (an) electron(s) and the more reactive the nonmetal will be. Nonmetals obtain a stable octet by gaining electrons. True or False? The lower the 1st IE, the more reactive a metal is. The easier the 1st electron is to lose, the more reactive the metal will be. This is because metals obtain a stable octet by losing electrons. As valence electrons are further from the nucleus and thus less tightly held it is easier to remove the first electron and IE decreases. Group: First IE decreases because as the ENC remains constant, the size of the atom increases and more shells are added to accomodate extra electrons. Period: As the ENC on the valence electrons increases and the atomic radius decreases, the electrons are more tightly held, closer to the nucleus, therefore, more energy must be put in to remove the first electron. 
Ionization Energy A(g) + energy → A+(g) + e Trends in Ionization Energy Increases from left to right, decreases from top to bottom. First Ionization Energy (IE) The energy required to remove the first electron from a neutral atom in the gaseous state. This ion has a greater atomic radius than that of its neutral atom because the gained electron repels from the other electrons. Anion A negatively charged ion frormed when an atom gains an electron.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
